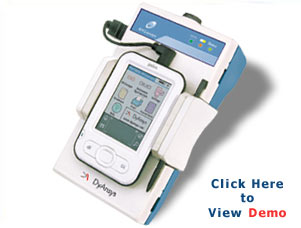
Portable ANSiscope
The Portable ANSiscope is a safe and non-invasive monitoring device for the Autonomic Nervous System. It is battery operated, operates in real time and is ideal for operation in a physician's practice or in the field.
Measurement
- The initial ANSindex reading is displayed after 60 R-R intervals (approx 1 minute)
- Subsequent readings are taken with every heartbeat
- The display is updated with every heartbeat
Data acquisition
- Standard leads acquired: I, II, III, aVR, aVL, aVF, V1-6
- Lead and gain selection: User can dynamically select lead/gain during recording
- Digital sampling rate: 1666 samples/sec (0.6ms samples)
- Sampling resolution: 16 bit, bipolar
- Frequency response: 0.5 Hz to 40 Hz
- Filters
- High Pass: 0.5 Hz cutoff
- Low Pass: 40 Hz cutoff
- Notch filter: 50Hz, 60Hz for power interference
- Input dynamic range: 5 mV
- Common mode rejection: >100dB
- DC offset capability: 300 mV DC
- Input lead impedance: >20M ohm at DC
Patient information
- Input at the start of the measurement
Treatment notes
- Can be appended during the measurement
Help facility
- Is available at any time from the display
Data display
- Choice of colour or monochrome displays
ANS monitor
- The ECG signals are displayed in real-time on the LCD Screen of the Portable ANSiscope
- The display also shows
- ECG lead selection
- Heart rate
- The sympathovagal balANS (from –50 to +50)
- Both, sympathetic and parasympathetic ANSi indices (from –50 to +50)
Measurement of autonomic dysfunction
- The display shows
- The static balANS™
- The percentage of autonomic dysfunction
- The diabetic autonomic neuropathy (DAN/CAN) group
- • Healthy • Early • Late • Advanced • Most Advanced
Data storage capability
- USB memory keys (Thumb drives) can be used for data
- storage/backup
- Also supports SCSI compatible USB mass storage devices
- Data can be exported to a PC via a USB memory key
Printing capability
- Prints on A4/letter size papers providing an inexpensive printing solution
- Works with HP C 5368 printer
- Regulatory compliance
- IEC 60601-1, IEC 60601-1-1, IEC 60601-1-2, IEC 60601-1-4, IEC 60601-2-27
- FDA Approved
Patient safety
- Patient leakage current: <10 A
- Classified as Class II - Type CF device as per IEC requirements
Product test and calibration
- Auto calibration at startup
- Extended tests and calibration can be performed including the ECG cable by connecting the ECG cables to calibration terminals provided in the product
Power specifications
- AC adapter input: 100-240V AC, 50/60 HZ
- AC adapter output: 12V DC @ 2amps Max
- Power consumption: <15W
Physical specifications
- Weight: <0.4 Kgs (including the battery)
- Dimensions: 175 x 95 x 112 (mm)
Environmental specifications
- Operating temperature: 0 to 45C
- vStorage temperature: –20C to +65C
- Humidity: 10% to 95%
- Pressure: 700 -1060 millibars
- Protected from patient defibrillation
Battery and charger
- Battery pack type: NiMH rechargeable battery
- Battery voltage: 3.6 VDC
- Battery capacity: 2300 mAh
- Typical battery operational time on full charge: >7 hours (without any peripherals connected)
- Typical battery charge time: 6 hours
- Auto shutdown time: 5 minutes
Standard accessories
- Print Tool, Silence Poster
- AC power adaptor with power cord
- Stylus and charger for display
- Cables
- USB interface cable for display
- 5 lead ECG cable
- Dyansys Disposable Surface electrodes
- roduct user manual
- Quick reference guide